Microfluidic Chain Reaction
Mohamed Yafia,1,2,† Oriol Ymbern,1,2,† Ayokunle O. Olanrewaju1,2, §, Azim Parandakh1,2, Ahmad Sohrabi Kashani1,2, Johan Renault1,2, Zijie Jin1,2, Geunyong Kim1,2, Andy Ng1,2, David Juncker1,2,* 5 1. Biomedical Engineering Department, McGill University, Montreal, QC, Canada 2. McGill Genome Centre, McGill University, Montreal, QC, Canada †Equal contribution. §Current address: Mechanical Engineering Department, University of Washington, Seattle, WA, USA.
Abstract
Chain reactions are characterized by initiation, propagation and termination, are stochastic at microscopic scales and underlie vital chemical (e.g. combustion engines), nuclear and biotechnological (e.g. polymerase chain reaction) applications.1–5 At macroscopic scales, chain reactions are deterministic and limited to applications for entertainment and art such as falling domino and Rube Goldberg machines. Appositely, microfluidic lab-on-a-chip (also called 15 a micro total analysis system),6,7 which were envisioned pursuant to microelectronic integrated circuits, are generally not integrated on a chip owing to an enduring dependency on cumbersome connections, peripherals, and on computers for automation.8–11 Capillary microfluidics integrate energy supply and flow control onto a single chip by using capillary phenomena, but programmability remains rudimentary with at most a handful (eight) operations possible.12–19 20 Here we introduce mesoscopic microfluidic chain reactions (MCRs) based on capillary phenomena for reliable programming and automation of complex liquid handling algorithms integrated in a chip. MCRs are encoded into the chip microarchitecture, 3D printed as a monolithic circuit, and deterministically propagated by the free-energy of a paper pump. With MCR, we sequentially triggered the release of 300 aliquots across chained, interconnected chips, 25 and automated a protocol for SARS-CoV-2 antibodies detection in saliva with visual and quantitative results by cell phone imaging. We automated and miniaturized for the first time the labor-intensive thrombogram with serial and parallel operations including timers and iterative cycles of synchronous flow and stop-flow sequences. Thrombograms with normal, hemophilia-like, and anticoagulant-spiked plasma were generated. MCRs are generalizable, and both the 30 density and number of chain reaction units are scalable. MCRs are untethered from and unencumbered by peripherals, encode programs structurally in situ, and form a frugal, versatile, 2 bona fide lab-on-a-chip with wide-ranging applications in liquid handling and point-of-care diagnostics.
Results
MCR encode the deterministic release of reagents stored in a series of reservoirs, with the release of reservoir n being conditional on emptying (draining) of the reagent in reservoir n-1, 5 and emptying reservoir n in turn triggering the release of reservoir n+1. Capillary domino valves (CDVs) encode this condition, and serially connect, i.e. chain, the reservoirs, and thus control the propagation of the chain reaction1 (Fig. 1a). MCRs were implemented in 3D-printed circuits using a common stereolithography printer with feature size from 100 μm to 1.5 mm, hydrophilized using a plasma (see Supplementary Fig. S1 and S2), sealed with a plain cover, and 10 connected to a capillary pump made of paper (filter papers or absorbent pads). The paper is spontaneously wetted by aqueous solution drawn from the microfluidic circuit by releasing free energy stored in the paper surface, and drives the chain reaction; expressed differently, the capillary pump generates a negative capillary pressure that is hydraulically propagated back into the circuit via the main channel and serially drains side-reservoirs connected via a small conduit, 15 called functional connection (further described below). CDVs form air links between adjacent reservoirs, serially connecting them along a path parallel to the main channel, but with filled reservoirs forming a liquid plug between CDV air links. When each reservoir empties, the length to the continuous air link increases from an air vent up to the next filled reservoir in the MCR, (Fig. 1a-d, and Video S1). This simple design structurally encodes the sequential release of an 20 arbitrary number N reservoirs without peripheral connection or moving parts.
MCR require ancillary capillary microfluidic components that fulfill different functions depending on the intended operation (e.g. loading, holding, mixing, and draining liquids following the MCR progression) to form fully integrated and scalable capillaric circuit (CCs). CCs are designed based on a library of building blocks including capillary pumps, flow 25 resistances, and many types of capillary valves (stop v., trigger v., retention v., retention burst v.) 13,14, and thus analogous to microelectronic integrated circuits, but lacking scalability and functionality theretofore. In MCRs, samples are loaded by capillary flow via an inlet with a capillary retention valve and entirely fill the reservoirs lined with three stop valves, including two with a dual retention burst valve function connecting to the two lateral CDVs, and one at the intersection of the functional connection and the main channel (Fig. 1c). Although the functional connection is a deceptively simple straight channel, it fulfills five key functions. It is (i) the air vent during filling of the reservoir, and (ii) a stop valve preventing the reagent from spilling into the main channel while it is empty. After filling of the main channel, it forms a (iii) hydraulic 5 link propagating the pressure from the main channel into the reservoir. (iv) It becomes the outlet and a flow resistance (discussed further below) during reservoir emptying, and (v) a capillary retention valve stopping air from invading the main conduit after the reservoir is emptied. As a result, many trade-offs guide its design.
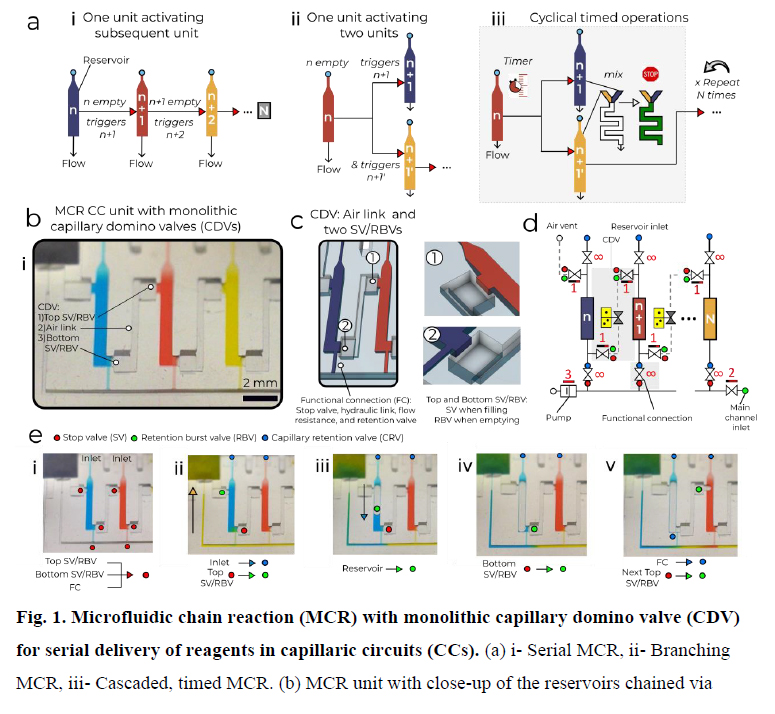
CDVs. (c) Close-up of CDV design: Air links form a pneumatic connection and capillary stop valves keep liquids out. (d) Symbolic view of the MCR unit with CDV (grey overlay) that includes an air link, two stop valves (SVs) and retention burst valves (RBVs). (e) Screen shots of Video S1 showing sequential steps for MCR where most of the capillary elements have dual functions that change upon either loading or during MCR. i- A loaded chip where the liquids 5 stop filling at different locations. ii- MCR is triggered (the inlet becomes a capillary retention valve and the top SV becomes a RBV). iii- Emptying of the first reservoir upon bursting of the top RBV. iv- The bottom SV momentarily becomes an RBV that bursts immediately. v- Air now occupies the emptied reservoir. The functional connection (FC) becomes a retention valve preventing the air from penetrating into in the main channel. The air link connects the air to the 10 RBV of the next reservoir, which bursts, and triggers reservoir emptying.
We sought to understand the design window and failure modes of MCR, notably under which conditions downstream of CDVs might trigger prematurely, using both theory and experiments. We analyzed the MCR based on an electrical analogy.20 Successful and incremental propagation of the MCR is conditional on preventing the breach of the liquid in reservoir n into 15 the CDV and air link connecting n+1, which is equivalent to stating that all the liquid must flow exclusively through the functional connection. This condition is satisfied when (see supplementary information for detailed derivation):
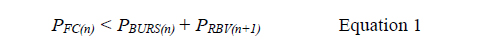
with PBURS being the leakage pressure of the stop valve, PRBV the retention pressure of the 20 retention burst valve at the extremities of the air link (and CDV) connecting reservoir n and n+1, respectively. We then derived PBURS (numerically) and PRBV (analytically) for the 3D printed CCs, and measured them for confirmation, Fig. 2b. Next, several MCRs featuring functional connections with large, and increasing RFC were tested with pumps with different capillary pressure and flow rates. The interplay between the resistance and the flow rate determines the 25 operational window for the CDV while they are inversely proportional. We found excellent concordance between theory and experiments for the operation window of the MCR, and failure only occurred for the highest values of RFC (no. 5 and 6), and only for the most powerful capillary pumps, Fig. 2c,d. The MCR designs used in the applications shown below, are well within the failure threshold and guarantee the reliability and robustness of the MCR.
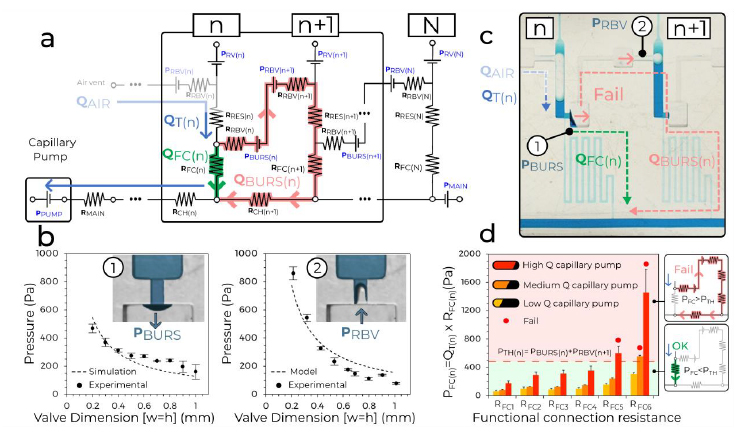
Fig. 2. Circuit analysis and experiments identify operational window for MCRs. (a) The equivalent electrical circuit of the MCR units shown in Fig. 1. (b) The burst pressure (1) and retention pressure (2) for a valve with different dimensions fitted with a numerical and an analytical model, respectively. Error bars are standard deviations from 3 experiments. (c) 5 Illustration of failure for a CDV where liquid leaks inside the air link leading to draining of reservoir n+1 prematurely. (d) Testing 6 MCRs with increasing RFC and three different paper pumps to determine the effect of varying the flow rate (n=3 for each paper pump and RFC). As predicted, the CDVs fail when the pressure drop across the functional connection PFC(n) exceeds the CDV threshold pressure PBURS(n) + PRBV(n+1).
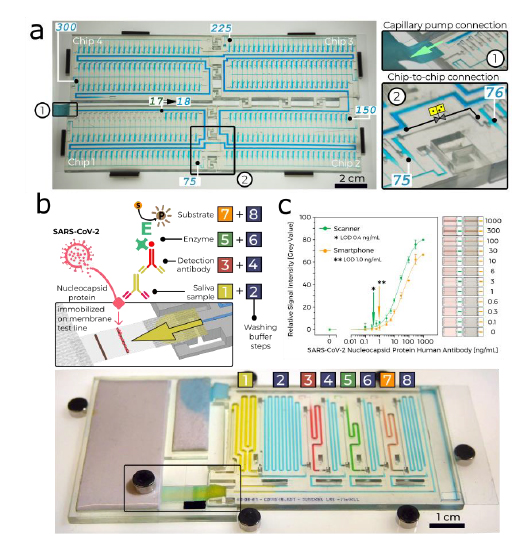
Fig. 3. Large-scale MCR and quantitative COVID-19 serology test. (a) A MCR of 300 aliquots stored in 4.9 μl reservoirs across four chained and interconnected chips. See Video S2. (b) Proof-of-concept diagnostic test for SARS-CoV-2 antibodies in saliva comprising two clickable chips, one with paper pumps and membrane, and one with sample, reagents and 5 washing buffers. Sequential, preprogrammed release via MCR is triggered by click-connecting chips and paper pump as detailed in the step-by-step diagram, assay schematic (left), and chip screenshot (bottom), see Video S3. The MCR supplies 4 reagents and 4 buffers in sequence. The functionality includes standard delivery and removal (by flushing) of solutions, individual metering via reservoir size (40 μl – 200 μl), flow speed and time control via the flow resistance 10 of the functional connection and the capillary-pressure of the paper pump. The enzymatic amplification produces a brown precipitate line visible to the naked eye. (c) Assay binding curve obtained by spiking antibody into saliva, and imaging by scanner and cell phone with representative images of the detection zone for each concentration, indicating the potential for quantitative point-of-care assays. Error bars are standard deviations of 3 replicate measurements for concentrations of 0 – 10 ng/ml, and of 2 replicate measurements for concentrations of 30 – 300 ng/ml
We designed a chip-to-chip interface with a leakage-free connection for liquid (main channel) and air (connecting the CDVs), respectively, and connected 4 chips with 75 MCR each, Fig. 3a and Video S2. This result demonstrates integrated, large scale fluidic operations by 5 ‘passive’ capillary microfluidics, beyond the capability of many active microfluidic systems
Automated COVID-19 saliva antibody test
We developed a point-of-care test for detecting antibodies against SARS-CoV-2 in saliva. The MCR drives a sequence of 8 steps commonly used in laboratory enzyme linked immunosorbent assay (ELISA), Fig. 3b. Conventional lateral flow assays with pre-dried reagents 10 cannot implement rinsing steps or enzymatic amplification, and accurate readout is limited to within a short time-window. Here, we used 3,3'-Diaminobenzidine as a substrate which upon enzymatic conversion produced a brown, persisting precipitate that could serve both as an immediate readout and a record for archival. The assay result can be visualized by naked eye, or quantified using a scanner or a smartphone integrated with a simple folded origami box to 15 minimize light interference, with a sensitive, quantitative, and reproducible output. (Fig. 3c and Supplementary Fig. S5).



